Recently, at an academic forum held by Shanghai Jiao Tong University, experts comprehensively reviewed the development of China's medical device industry, and discussed the future development direction of the medical device industry and the problems that need to be addressed. As a guest expert, Cai Tianzhi, Director of Medical Device Department of China Chamber of Commerce for Imports and Exports of Medicines and Health Products, shared his understanding of these issues during a three-hour lecture. This article extracts some of the content for peer discussion.
As a booming market demand sunrise industry, the medical device industry in recent years has a good momentum of development. In 2007, the output value of medical devices in China's GDP accounted for 0.24%; In 2011, it increased to 0.29% -- although the proportion is not large, it has been on the rise. In 2012, the output value of China's medical device industry is expected to exceed 170 billion yuan.


Change - technology and quality improvement
In recent ten years, the technical structure and product quality of medical devices in China have changed significantly, especially in the fields of patient monitoring products, medical imaging equipment, clinical laboratory equipment and minimally invasive interventional therapy products. In addition, a number of innovative products with completely independent intellectual property rights have emerged in China, such as stereotactic ultrasound focused treatment system, excimer laser human eye aberration correction system, body rotating gamma knife, and sleep monitoring system.
Change two: the number of firms has soared
The number of medical device manufacturers in China is increasing year by year: 14,337 in 2010, among which there are 4,015 first-class medical device manufacturers, 7,906 second-class medical device manufacturers, 2,416 third-class medical device manufacturers and 1,863 national and provincial key supervised enterprises. By the end of November 2012, the number of medical device manufacturers in China had rapidly increased to more than 16,000, with state-owned enterprises, foreign-funded enterprises, joint ventures and private enterprises accounting for 3%, 37% and 60% respectively. Among them, there are more than 200 enterprises with an output value of over 100 million yuan; There are less than 20 enterprises with an output value of over 1 billion yuan, and foreign-funded enterprises and joint ventures account for the vast majority. Statistics in 2011 show that foreign investors and Hong Kong, Macao and Taiwan investment enterprises constitute the main force of China's medical device industry, their assets accounted for 43% of the total assets of China's medical device industry.
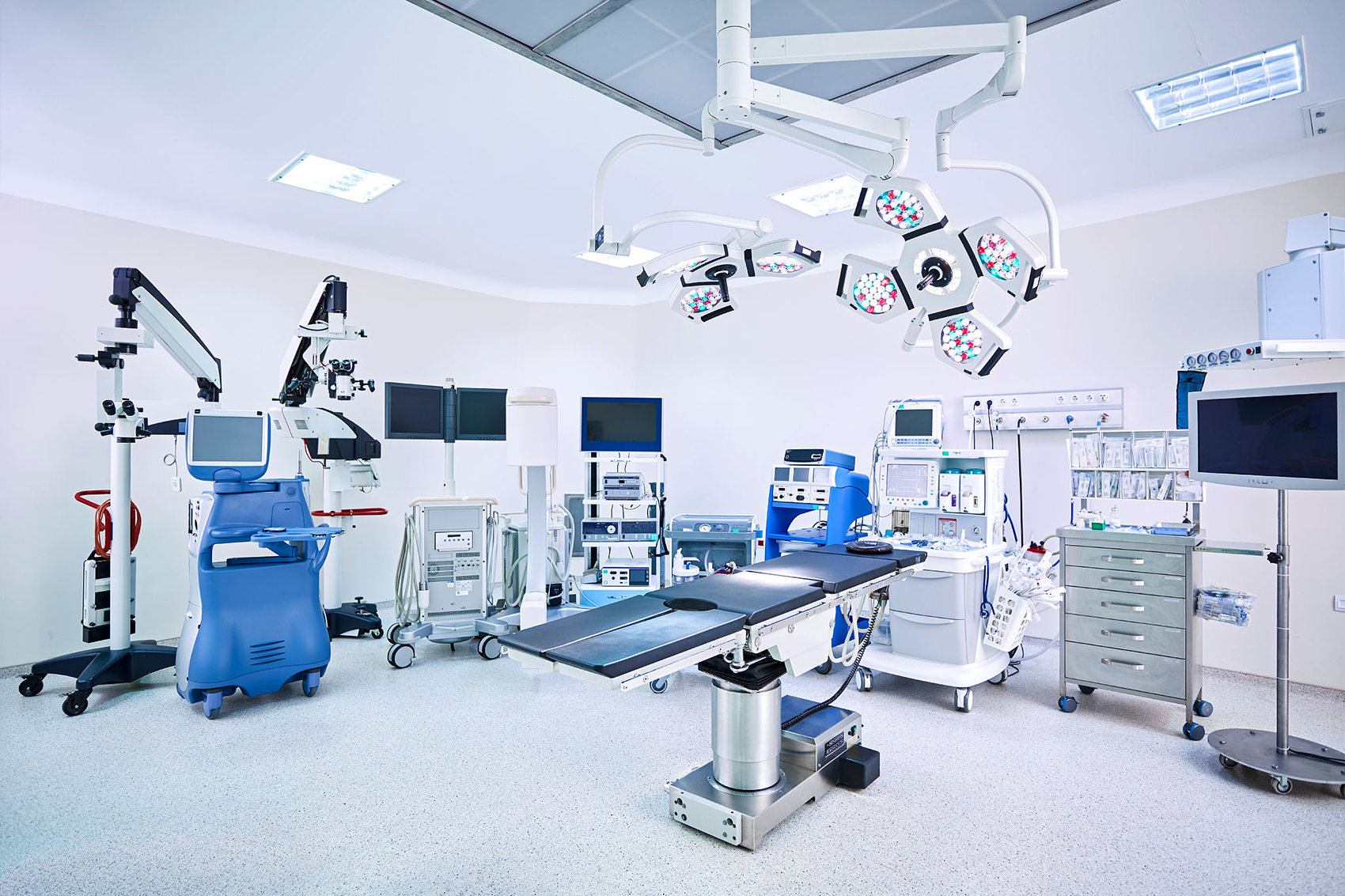
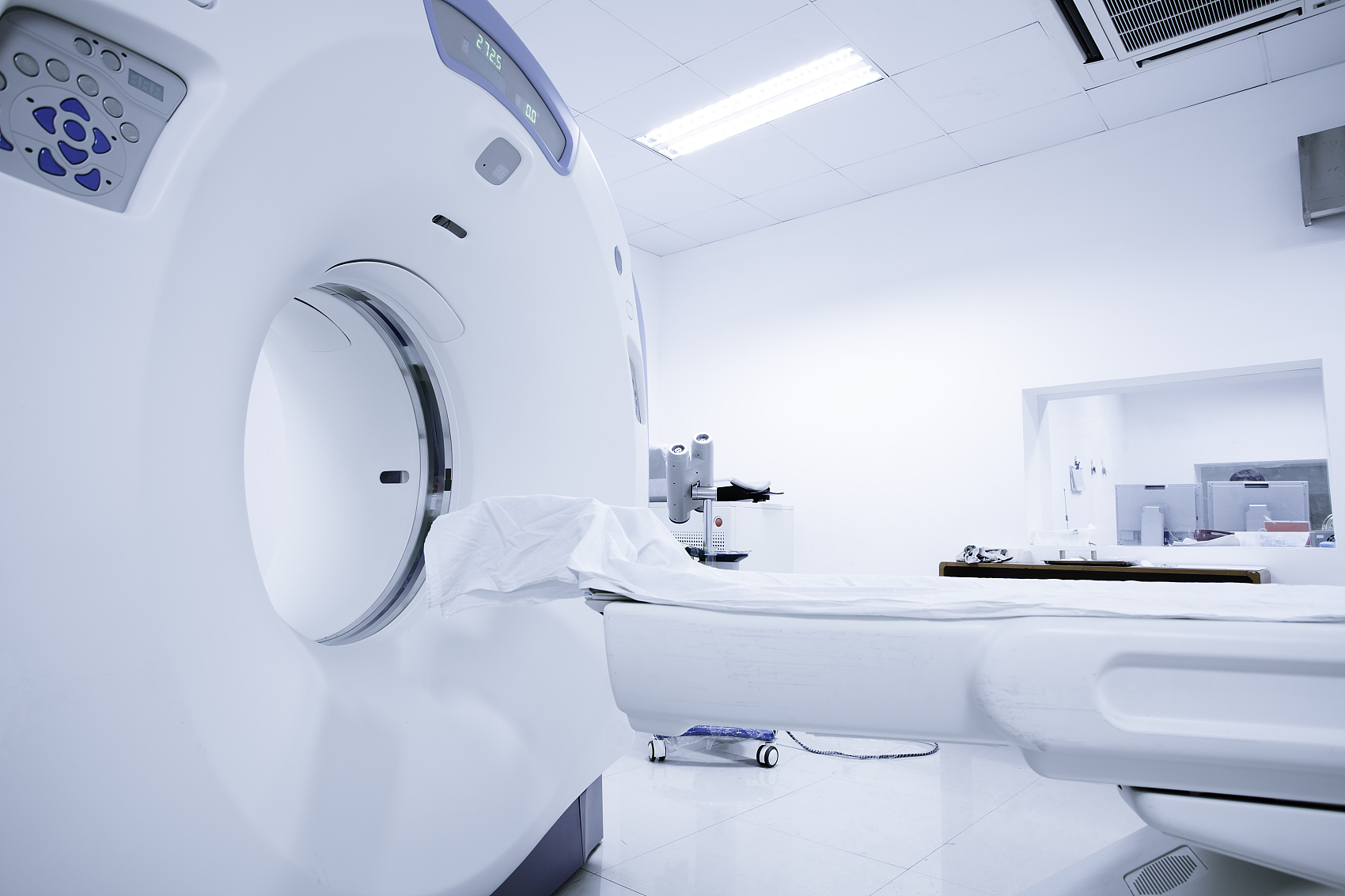
Changes in the three: regional advantages highlighted
The three regions of Pearl River Delta, Yangtze River Delta and Bohai Bay have gradually developed into three clusters of China's medical device industry by relying on their industrial technology, scientific and technological talents, clinical medical foundation and policy advantages. The sum of the output value of medical devices in these three regions and the sum of the sales value of medical devices account for more than 80% of the output value and total sales value of medical devices in China.
The common points of these three regions are as follows: they have a relatively strong industrial foundation for medical equipment support, as well as a relatively strong industry for electronics, machinery and equipment manufacturing and chemical industry, making it convenient to carry out technical cooperation and product matching; Convenient transportation at home and abroad, with a number of international level ports and docks, conducive to the development of technological exchanges and trade; Institutions of higher education and talents with high knowledge and high technology are relatively concentrated, and there are strong potential human resources and technological development potential. The level of medical scientific research and clinical medicine is higher than that of other regions, and there are a large number of tertiary hospitals. The clinical trial research and clinical evaluation ability of new medical devices is very strong. There are strong financing channels, including state-owned capital, private capital and foreign capital.
The three regions have their own characteristics
The Yangtze River Delta and the Bohai Bay are trans-provincial large regions, and their original medical device manufacturing industry has a good foundation. After adjustment and reorganization, they have the characteristics of combining new and old; The Yangtze River Delta has developed township enterprises, Jiangsu has a strong organic chemical industry, disposable medical supplies industry development outstanding; The Pearl River Delta is a part of Guangdong Province. Although the original medical equipment manufacturing industry is poor, its modern medical equipment manufacturing industry benefits from the reform and opening up policy and the introduction of foreign capital, as well as the rapid development of home appliances and electrical manufacturing industry, so it has become a latecomer in the medical equipment industry. Although the output value of medical devices in this region is lower than that of the Yangtze River Delta and Bohai Bay, from the perspective of the technical structure of medical devices and the technical added value of the main products, Shenzhen in the Pearl River Delta ranks the first place in China and is a pearl in the national medical device industry.
In 2011, the top five regions of medical device sales revenue were Jiangsu, Guangdong, Shandong, Shanghai and Beijing, whose sales revenue accounted for 64.16% of the total. Among them, Jiangsu and Guangdong accounted for 23.16% and 15.87% respectively. The top five regions in terms of sales profit were Jiangsu, Guangdong, Beijing, Shanghai and Shandong, which accounted for 23.51%, 19.28%, 11.91%, 7.99% and 7.70% respectively, with a total of 70.39%. This shows that these five regions are the important distribution regions of the medical device industry in China, with absolute advantages in terms of scale and profit. In addition, the product technology level and added value of Beijing, Chongqing, Guangdong, Zhejiang and Sichuan are obviously higher than those of other provinces (cities), so the profit margin of medical device sales is also higher.
Change in the fou:mergers and acquisitions to accelerate restructuring
It is a major trend in the medical device industry for enterprises to focus more on a certain segment or take the road of diversified development through merger and reorganization. Since 2010, mergers and acquisitions in China's medical device industry have accelerated significantly. These mergers and acquisitions followed three patterns: 1. Focus on market segments and achieve leading segments; 2. 2. To become a comprehensive giant through mergers and acquisitions; 3. Realize the integration of medical electricity with the help of scientific and technological advantages. For example, Shenzhen Mindray, Weigao shares, fish diving medical, Lepu medical have diversified through joint ventures, mergers and acquisitions, and become the leader of integration; Minimally Invasive Medical, Comfort Medical, Tronson Medical, Jiuan Medical, Yangpu Medical and Xinhua Medical are the leaders in the field of segmentation.
Post time: Jul-01-2021
